Does atomic size increase down a group?
4.5 (515) · $ 6.50 · In stock
Does atomic size increase down a group?
Does atomic size increase down a group

Atomic Radius Trend, Periodic Table
:max_bytes(150000):strip_icc()/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
Size of the Elements on the Periodic Table

1.1 Exercise 4 - Ionisation Energies
How does atomic size vary on moving across a period from left to right and in a group, top to bottom? Why? - Quora

Solved Question 1 Fill in the blanks by select Going down a

Understanding Atomic Radius Trends: The 2 Key Principles

6answer Module Sk015 Chapter 3 1 .pdf - Practice Module Chapter 3

Atomic Structure and Periodic Trends, PDF

Why does the atomic size increases down a group? - Quora

1.1 Exercise 4 - Ionisation Energies
Why does atomic radius decrease as you go across a period?
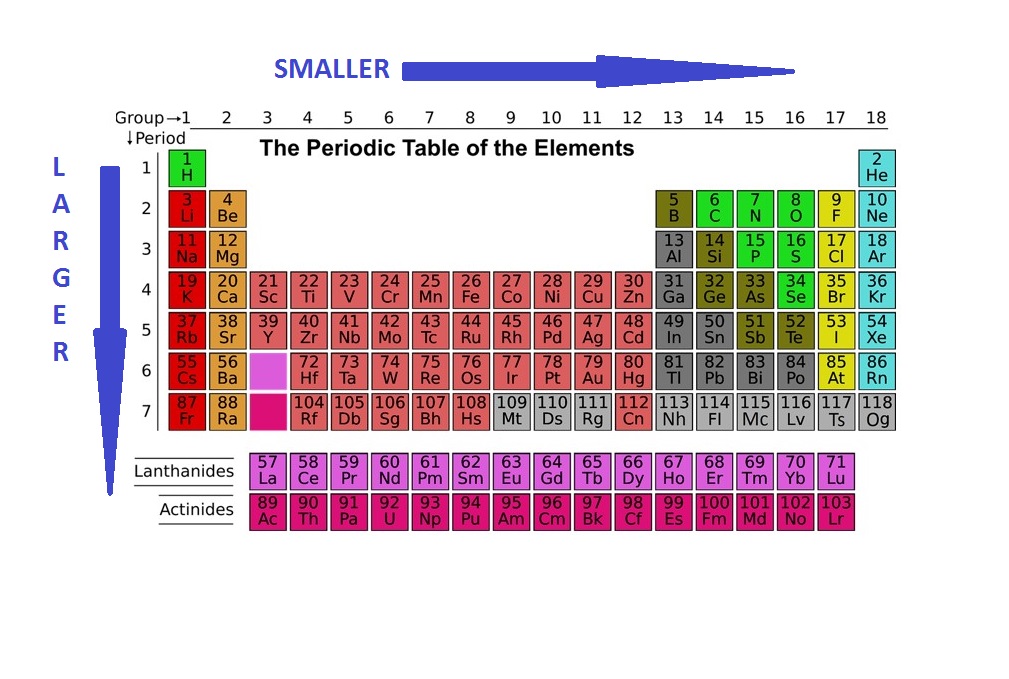
Understanding Atomic Radius Trends: The 2 Key Principles

Section 4.5—Periodicity Objectives: Define periodic trend - ppt download

Periodicity 3: Why Atomic Radii Increases Down a Group
What are the periodic trends for atomic radii, ionization energy, and electron affinity?