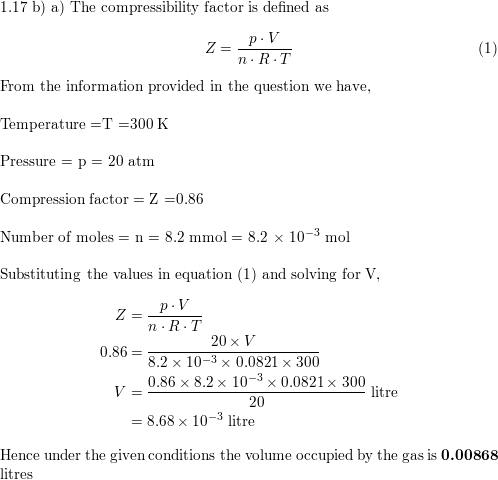- Home
- compressibility factor equation
- The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is
The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is
4.7 (293) · $ 9.99 · In stock
Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor compressibility factor for onemole of a van der waals gas at 0c
Click here👆to get an answer to your question ✍️ The compression factor -compressibility factor- one mole of a van der Waals gas 0-C and 100 atm pressure is found to be 0-5- Assuming that the volume of a gas molecule is negligible- calculate the van der Waals- constant a

Chapter 01 The Nature of Physical Chemistry, PDF, Gases

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application

Random, PDF, Gases

02 mole of a van der Waals gas pressure of 0.1 alin. Civanges unpredictably (B-16. What is the compressibility factor (Z) 0.02 mole of a Assume the size of gas molecules is

Thermodynamics: An Engineering Approach - 5th Edition - Part II by

Calculate pressure exerted by 1 mole of a van der Waal's gas at a temp
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect

The compression factor (compressibility factor) for one mole of a Van der..

Non-ideal behavior of gases (article)

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics

jo 22] What is the compressibility factor (Z) 0.02 mole of a van der Waals' gas pressure of 0.1 atm. Assume the size of gas molecules is negligible.

The compression factor (compressibility factor) for 1 mol of a van der